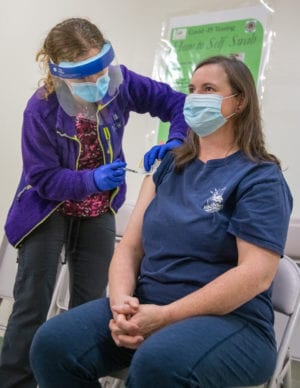
Three health care workers became the first Cordovans vaccinated against the novel coronavirus Wednesday, Dec. 16.
Kathleen Kroll, a registered nurse, Kelly Kedzierski, Cordova Community Medical Center’s chief nursing officer, and Jeff Bailey, a radiology technician, were first in line at CCMC’s ambulance bay to receive a shot of Pfizer-BioNTech coronavirus vaccine flown in from Anchorage earlier that evening. Kroll, Kedzierski and Bailey were among five people vaccinated Dec. 16.
The vaccine gave Kroll, who spends her workdays performing COVID-19 tests, a greater sense of security, she said.
“It protects me, because I’m exposed to everybody that comes in,” Kroll said. “But it also protects everybody I work with. If you’re coming into the emergency department because you broke your arm, the last thing you want is me to breathe COVID on you… I’m protecting patients as well as myself.”
CCMC’s supply of Pfizer-BioNTech’s vaccine will first be administered to medical staff, first responders and long-term care residents, in accordance with state guidelines. Ilanka Community Health Center will soon receive doses of a coronavirus vaccine developed by Moderna, city officials said. Both vaccines require two doses, spaced about three weeks apart, to achieve maximum effectiveness, according to a release from the Centers for Disease Control and Prevention.
Alaska began receiving more than 35,000 doses of coronavirus vaccine Sunday, Dec. 13 for distribution across the state. Of those doses, 11,700 were earmarked for Alaska Native Tribes by the Indian Health Service, according to Associated Press reports. The city of Cordova will provide updated information as a wider pool of Cordova residents becomes eligible for the vaccine, according to a city announcement.
How do mRNA vaccines work?
Messenger ribonucleic acid, or mRNA, is a molecule that conveys genetic information. While most vaccines use a weakened form of a disease, mRNA vaccines create immunity without exposing the patient to a disease at all.
Coronavirus mRNA vaccines, like those manufactured by Pfizer-BioNTech and Moderna, deliver mRNA containing instructions to produce part of a protein that is unique to the novel coronavirus. Though the protein fragment is not infectious, it allows the patient’s body to “recognize” the same protein when coronavirus particles enter the system, triggering an immune response, according to a CDC release. The mRNA in a vaccine does not enter the cell nucleus and cannot alter a patient’s genetic makeup.
Although the coronavirus vaccination effort is the first time an mRNA vaccine has been publicly distributed in the U.S., clinical trials of mRNA vaccines for influenza, Zika virus and rabies have been carried out in the past. Scientists hope that, in the future, mRNA vaccine technology will allow one injection to provide immunity against multiple diseases, according to a CDC release. Some researchers have also used mRNA to trigger the immune system to attack specific cancer cells.

A slim risk of allergic reaction
As she prepared to receive the vaccine, Kroll said she wasn’t worried about the possibility of an allergic reaction, as such reactions are both treatable and rare.
“I really believe they’ve put together a vaccine that can be trusted,” Kroll said. “They’ve given it to over 50,000 people with great success — very few side-effects.”
A Juneau health care worker experienced a severe allergic reaction to the vaccine Dec. 15, the AP reported. The worker, who reported feeling flushed and short of breath less than 10 minutes after receiving the shot, was kept overnight at Bartlett Regional Hospital. She had recovered by the next day, hospital officials said.
Unlike two British health care workers who had similar reactions to the vaccine during the prior week, the Juneau worker had no history of allergic reactions. Despite her ordeal, the worker remained “enthusiastic” about having taken the vaccine, Bartlett Regional Hospital Emergency Room Medical Director Dr. Lindy Jones told the AP.
Dr. Anne Zink, Alaska’s chief medical officer, said the state doesn’t plan to alter its vaccine distribution.
“This is all kind of part of what we’ve been looking for and expecting,” Zink told the AP.
Dec. 13, the CDC issued a statement saying that people who have previously experienced severe reactions to vaccines or injectable drugs can still receive the Pfizer-BioNTech vaccine, but that they should be monitored for 30 minutes afterward. This amended a previous CDC statement recommending that people who have experienced severe reactions not receive the vaccine.
Rare severe allergic reactions have also been observed with other, widely distributed vaccines. A 2015 study of individuals vaccinated for polio, measles, meningitis and other diseases found the rate of severe allergic reactions was about one case per one million doses, according to AP reports. A Pfizer-BioNTech study of 42,000 people found the rate of allergic reaction was roughly the same among those who received the company’s coronavirus vaccine as among those who received a placebo injection. A U.S. Food and Drug Administration review of the Pfizer-BioNTech study found that 0.63% of vaccine recipients reported some degree of allergic symptoms, compared to 0.51% of the placebo group.
“It’s just so high-profile that every little thing that happens all the time is going to get magnified,” Dr. Ashish Jha, dean of Brown University’s school of public health, told the AP. “We should talk about it, we should be honest with people, but we should put it into context and help people understand… There is a small proportion of people who have an allergic reaction to almost any medicine.
A sore spot where the shot was given, or a low-grade fever, are common side effects of many vaccines and can be expected to go away on their own after a few days, according to a CDC statement.














